institute for continuous health transformation
(InHealth)
Joaquim Cardoso MSc
Founder and Chief Researcher & Editor
January 29, 2023
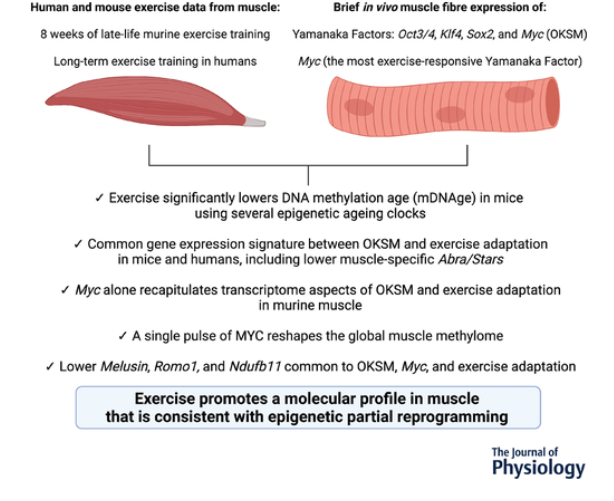
KEY MESSAGES
A new study investigates how exercise can help rejuvenate aging muscles.
- Doctors call physical exercise a “polypill,” because it can prevent and treat many of the chronic diseases that are associated with aging.
- A new study of muscle fibers from mice and humans shows how exercise affects gene expression.
- The exercise-induced changes “reprogram” the epigenetic expression of the fibers to a more youthful state.
- The findings could provide leads for the development of drugs to mimic these benefits in people who are unable to exercise.
INFOGRAPHIC
DEEP DIVE

Can exercise reverse muscle aging? Yes, and this is how
Medical News Today
James Kingsland , Fact checked by Hannah Flynn
January 24, 2023 —
Research shows that people who exercise regularly not only strengthen their muscles but also improve their overall health, regardless of how late in life they start.
- For example, recent studies have found that exercise reduces the risk of cardiovascular disease,
- as well as Alzheimer’s and Parkinson’s in older people.
- Conversely, reductions in muscle mass and strength are associated with lower quality of life and higher mortality from all causes.
As a result of its proven ability to prevent and treat several chronic diseases at low cost, doctors have called exercise a drug-free “polypill” that can benefit nearly everyone.
“Exercise is the most powerful drug we have,” says Dr. Kevin Murach, assistant professor at the Exercise Science Research Center, University of Arkansas, Fayetteville, AR.
He believes that exercise should be considered a health-enhancing, potentially life-extending treatment, alongside medications and a healthy diet.
“Exercise is the most powerful drug we have…
exercise should be considered a health-enhancing, potentially life-extending treatment, alongside medications and a healthy diet.

Scientists hope that a better understanding of how exercise rejuvenates aged muscle at a molecular level will provide clues for future anti-aging therapies.
Exercise may turn back the clock in muscle fibers by promoting the “epigenetic reprogramming” of chromosomes in the cells’ nuclei.
Epigenetics refers to how chemical changes affect the activity or “expression” of genes.
For example, proteins called transcription factors can dial up the expression of particular genes when they bind to specific DNA sequences.
In 2012, Dr. Shinya Yamanaka shared the Nobel Prize for Medicine for his discovery that four transcription factors can revert specialized, mature cells to more youthful, flexible cells called pluripotent stem cells.
The four factors are called Oct3/4, Klf4, Sox2, and Myc, or OKSM for short.
In 2012, Dr. Shinya Yamanaka shared the Nobel Prize for Medicine for his discovery that four transcription factors can revert specialized, mature cells to more youthful, flexible cells called pluripotent stem cells.
The four factors are called Oct3/4, Klf4, Sox2, and Myc, or OKSM for short.

In a new study whose results appear in The Journal of Physiology, Dr. Murach and his colleagues compared the effects of OKSM factors on gene expression in the muscle fibers of mice …
…that had access to an exercise wheel, and mice that had no access.
In addition, they compared the effects of OKSM factors on muscle with the effects of a single transcription factor, Myc. Scientists have found that exercise induces the expression of Myc to a greater extent than the other three factors.
The researchers also investigated how exercise alone affected gene expression in muscle fibers from both mice and humans.
The mice were 22 months old, which is equivalent to a human age of around 73 years.
Mice in the exercise group were free to run on an unweighted wheel for the first week, then, over the next 8 weeks, the scientists made the wheel progressively heavier by attaching magnetic weights to it.
The results suggest that exercise reprograms muscle fibers to a more youthful state through increased expression of the genes that make Yamanaka factors, in particular Myc.
The results suggest that exercise reprograms muscle fibers to a more youthful state through increased expression of the genes that make Yamanaka factors, in particular Myc.
Dr. Murach suggests that the findings may one day lead to the development of drugs that supercharge the exercise response of muscles in people who are confined to bed, or the muscles of astronauts in zero gravity.
… the findings may one day lead to the development of drugs that supercharge the exercise response of muscles in people who are confined to bed, or the muscles of astronauts in zero gravity.

But he dismisses the idea of a pill that boosts the expression of Myc ever replacing the need to exercise.
For one thing, exercise has beneficial effects throughout the body, not just in muscle.
In addition, Myc has been linked to cancer, so there are inherent risks in artificially boosting its expression.
In addition, Myc has been linked to cancer, so there are inherent risks in artificially boosting its expression.

In their paper, the researchers also note that drugs that are gaining a popular reputation as “life-extending” may actually block some of the beneficial effects of exercise on muscle.
Dr. Murach told Medical News Today:
“Evidence suggests that ‘life-extending’ drugs such as metformin and rapamycin interfere with the positive benefits of exercise specifically in skeletal muscle.”
He said it was “not outside the realm of possibility” that the drugs could disrupt the epigenetic reprogramming of muscle that happens with exercise.

MNT asked exercise physiologists to recommend the best type of exercise for older people.
“For individuals over 70 I would highly recommend low-impact, full-body workouts with a focus on lower body and core,” advised John C. Loges, an exercise physiologist at eVOLV Strong.
“For individuals over 70 I would highly recommend low-impact, full-body workouts with a focus on lower body and core,” …
“Resistance training is not only suitable but highly recommended for those in their 70s and beyond,” he said.
“The key is starting slow and progressing slowly with consistency,” he added.
“[W]alking is an activity that I recommend, along with resistance and mobility training,” advised Melissa Hendrix Wogahn, an exercise physiologist at Joy of Active Living who offers fitness and health education for older adults.
She recommended strength training at least two days a week and mobility training, including stretching, every day.
“[W]alking is an activity that I recommend, along with resistance and mobility training, …
… strength training at least two days a week and mobility training, including stretching, every day.
“In terms of frequency, an older adult can walk every day, assuming they have no contraindications,” she added.
The authors of the new study acknowledge that it had some limitations.
For example, the type of exercise, training status, biological sex, and several other factors may affect gene expression changes associated with exercise.
In addition, they emphasize the importance of investigating the functional consequences of epigenetic reprogramming in skeletal muscle.
Originally published at https://www.medicalnewstoday.com on January 24, 2023.
ORIGINAL PUBLICATION

A molecular signature defining exercise adaptation with ageing and in vivo partial reprogramming in skeletal muscle [excerpt version]
The Journal of Phisiology (The Physiological Society)
Ronald G. Jones III, Andrea Dimet-Wiley, Amin Haghani, Francielly Morena da Silva, Camille R. Brightwell, Seongkyun Lim, Sabin Khadgi, Yuan Wen, Cory M. Dungan, Robert T. Brooke, Nicholas P. Greene, Charlotte A. Peterson, John J. McCarthy, Steve Horvath, Stanley J. Watowich, Christopher S. Fry, Kevin A. Murach … See fewer authors
19 December 2022
Abstract
- Exercise promotes functional improvements in aged tissues, but the extent to which it simulates partial molecular reprogramming is unknown.
- Using transcriptome profiling from (1) a skeletal muscle-specific in vivo Oct3/4, Klf4, Sox2 and Myc (OKSM) reprogramming-factor expression murine model; (2) an in vivo inducible muscle-specific Myc induction murine model; (3) a translatable high-volume hypertrophic exercise training approach in aged mice; and (4) human exercise muscle biopsies, we collectively defined exercise-induced genes that are common to partial reprogramming.
- Late-life exercise training lowered murine DNA methylation age according to several contemporary muscle-specific clocks.
- A comparison of the murine soleus transcriptome after late-life exercise training to the soleus transcriptome after OKSM induction revealed an overlapping signature that included higher JunB and Sun1.
- Also, within this signature, downregulation of specific mitochondrial and muscle-enriched genes was conserved in skeletal muscle of long-term exercise-trained humans; among these was muscle-specific Abra/Stars.
- Myc is the OKSM factor most induced by exercise in muscle and was elevated following exercise training in aged mice.
- A pulse of MYC rewired the global soleus muscle methylome, and the transcriptome after a MYC pulse partially recapitulated OKSM induction.
- A common signature also emerged in the murine MYC-controlled and exercise adaptation transcriptomes, including lower muscle-specific Melusin and reactive oxygen species-associated Romo1.
- With Myc, OKSM and exercise training in mice, as well habitual exercise in humans, the complex I accessory subunit Ndufb11 was lower; low Ndufb11 is linked to longevity in rodents.
- Collectively, exercise shares similarities with genetic in vivo partial reprogramming.
Key points
- Advances in the last decade related to cellular epigenetic reprogramming (e.g. DNA methylome remodelling) toward a pluripotent state via the Yamanaka transcription factors Oct3/4, Klf4, Sox2 and Myc (OKSM) provide a window into potential mechanisms for combatting the deleterious effects of cellular ageing.
- Using global gene expression analysis, we compared the effects of in vivo OKSM-mediated partial reprogramming in skeletal muscle fibres of mice to the effects of late-life murine exercise training in muscle.
- Myc is the Yamanaka factor most induced by exercise in skeletal muscle, and so we compared the MYC-controlled transcriptome in muscle to Yamanaka factor-mediated and exercise adaptation mRNA landscapes in mice and humans.
- A single pulse of MYC is sufficient to remodel the muscle methylome.
- We identify partial reprogramming-associated genes that are innately altered by exercise training and conserved in humans, and propose that MYC contributes to some of these responses.

METHODS & OTHER SECTIONS
See the original publication (this is an excerpt version_

RESULTS
Late-life exercise training associates with lower skeletal muscle mDNAge using contemporary muscle-specific epigenetic clocks
To corroborate our previously-described exercise training-mediated shift in methylation age toward a younger status in muscle, female C57BL/6N mice (n = 10) completed PoWeR from 22 to 24 months. Approximately 22-month-old mice are estimated to be equivalent to ∼73-year-old humans (Dutta & Sengupta, 2016). Age-matched untrained mice were sedentary controls (n = 9). Muscle methylation was quantified with the Horvath Mammalian Methylation Chip that focuses on conserved cytosines (Arneson et al., 2022) and analysed with contemporary murine mDNAge clocks (Mozhui et al., 2022). After PoWeR, skeletal muscle mDNAge was lower relative to sedentary using the Horvath Muscle Clock (−5.7 weeks, P = 0.04) and Developmental Muscle Clock (−7.1 weeks, P = 0.02) (Fig. 1A). The magnitude of epigenetic age mitigation by late-life exercise in muscle was similar to our previous report using an independent cohort of mice and a different mDNAge analysis (8 weeks, n = 5 per group, P = 0.07) (Murach et al., 2021). Running volume was slightly lower in the present study (4.3 km day–1 vs. 6.4 km day–1). Consistently lower mDNAge with exercise motivated us to compare transcriptome profiles of a partial reprogramming stimulus (OKSM expression) vs. exercise adaptation in muscle to identify a common gene expression signature.
Figure 1. Comparison of Oct3/4, Klf4, Sox2 and Myc (OKSM) expression to late-life progressive weighted wheel running (PoWeR) in mouse muscle
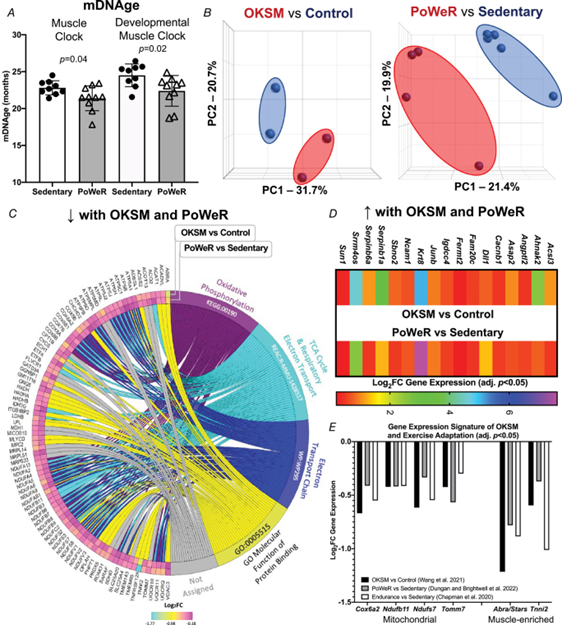
A, DNA methylation age (mDNAge) derived from clocks developed by the Horvath laboratory applied to late-life PoWeR (n = 10) and sedentary control (n = 9) mouse gastrocnemius muscle. B, PCA plots showing DESeq2-normalized gene count transcriptome data from OKSM expression (n = 2) vs. control (n = 2) in mouse soleus muscle fibres (Wang et al., 2021) and the late-life PoWeR (n = 4) vs. the sedentary (n = 5) transcriptome in soleus muscle (Dungan, Brightwell et al., 2022). C, chord diagram showing significantly downregulated genes common across OKSM and late-life PoWeR in the soleus. D, upregulated genes common across OKSM and late-life PoWeR in the soleus. E, downregulated genes common across OKSM, late-life PoWeR and long-term exercise training in humans (Chapman et al.) All data are reported as the mean ± SD.
The transcriptome resulting from OKSM expression in muscle fibres overlaps with the late-life exercise adaptation transcriptome in mouse muscle
RNA-seq data from the soleus muscle of adult mice after 2.5 days of genetic OKSM expression (Wang et al., 2021) was compared to RNA-sequencing data from the soleus of late-life PoWeR mice (Dungan, Brightwell et al., 2022). The soleus adapts more substantially to PoWeR than other hindlimb muscles in young and old mice (Englund et al., 2020; Murach et al., 2020), with similar gene expression patterns regardless of age (Dungan, Brightwell et al., 2022). The soleus also contains similar proportions of the primary myosin fibre types found in human vastus lateralis (Dungan, Brightwell et al., 2022; Murach et al., 2019, 2020), further justifying our focus on this muscle.
OKSM induction and PoWeR revealed stark effects on gene expression for each intervention (Fig. 1B). With OKSM, 412 genes were upregulated and 523 genes were downregulated (adjusted P < 0.05) (see Supporting information, Table S1). Pathway analysis of OKSM induction revealed repression of genes associated with oxidative phosphorylation, in agreement with the analysis from the Belmonte laboratory (Wang et al., 2021). We compared the list of genes downregulated by OKSM with those downregulated by PoWeR (377 genes, adjusted P < 0.05) (see Supporting information, Table S2). Approximately one-quarter of downregulated genes overlapped (87 genes) (Fig. 1C). Pathway analysis of shared downregulated genes revealed oxidative phosphorylation (KEGG, adjusted P = 9.77 × 10−54), the citric acid cycle (Reactome, adjusted P = 8.31 × 10−56) and electron transport chain (Wikipathways, adjusted P = 2.08 × 10−44) as the top pathways (Fig. 1C). The muscle-enriched genes fast skeletal muscle troponin (Tnni2) (Sheng & Jin, 2016) and striated muscle activator of Rho signalling (Stars, or Abra) (Arai et al., 2002; Wallace et al., 2012) were also lower with OKSM and PoWeR; this suggested a repression of muscle cellular identity, consistent with partial reprogramming effects. Seventeen upregulated genes were common between OKSM and PoWeR datasets (Fig. 1D), including the potent muscle mass-regulator JunB (Raffaello et al., 2010) and myonuclear anchor Sun1 (Lei et al., 2009). Perhaps alteration to Sun1 could be related to myonuclear shape changes that occur with exercise training in rodents (Battey et al., 2023; Murach et al., 2020; Rader & Baker, 2022). Collectively, our analysis unearthed a canonical skeletal muscle gene expression signature common between OKSM and exercise adaptation.
Gene expression alterations with OKSM and exercise adaptation in mouse muscle are conserved in human muscle after prolonged exercise training
To validate our murine findings in humans we queried an RNA-sequencing dataset from muscle biopsy samples of women with at least 15 years of mixed-modality endurance training history (n = 9, ∼42 years old) (Chapman et al., 2020). Comparing our murine PoWeR data to long-term training in humans seemed appropriate as 8 weeks of high-volume PoWeR corresponds to ∼10% of the mouse lifespan and could thus be characterized as prolonged training. Several mitochondrial-related genes downregulated by OKSM and PoWeR were significantly lower in muscle of long-term endurance-trained athletes relative to sedentary age-matched controls (COX6A2, adjusted P = 0.046; NDUFB11, adjusted P = 0.017; NDUFS7, adjusted P = 0.041; TOMM7, adjusted P = 0.028). Lower levels of mitochondrial genes with endurance training appears to be counterintuitive. To further explore this observation, we compared genes that were altered in female endurance trained muscle to the human MitoCarta 3.0 database (Rath et al., 2021). Although 100 genes that were higher with training overlapped with MitoCarta, 52 genes that were lower also overlapped; these included NDUFs such as NDUFAF3 and NDUFS7, as well as COX6A1 and COX6A2 (see Supporting information, Table S3). Thus, endurance training adaptations in skeletal muscle are not characterized by unanimously elevated mitochondrial gene levels. In addition to mitochondrial genes, muscle-enriched TNNI2 and ABRA (STARS) were lower in exercise-trained human muscle (adjusted P = 0.002 and 0.003, respectively), consistent with OKSM expression and late-life PoWeR in mice (Fig. 1E).
Myc is an exercise-responsive transcription factor in muscle that influences the gene expression landscape with PoWeR in aged mice
The OKSM genetic mouse model was designed to induce all four Yamanaka factors, but Klf4 gene expression was not increased significantly in the soleus in the OKSM experiment (Wang et al., 2021); we corroborate this in our analysis (1.05 Fold change (FC) OKSM vs. Sedentary, adjusted P = 0.95). All four factors can contribute to muscle adaptation via a change in activity independent of expression levels, but partial reprogramming was probably most attributable to Oct3/4, Sox2 and Myc. Of OKSM, only MYC is significantly induced by a bout of exercise in skeletal muscle from healthy humans according to a transcriptome meta-analysis (n = 64–187 subjects, acute resistance and endurance exercise) (Pillon et al., 2020) (Fig. 2A). It is worth noting that the KLF4 response is quite variable but may be induced in the first few hours after endurance exercise. Using up- and downregulated differentially expressed gene lists from our PoWeR RNA-seq data in the soleus, we performed Lisa analysis for insight on the potential contribution of MYC in the control of gene expression (Murach et al., 2022; Qin et al., 2020). Lisa incorporates transcriptome input data with an extensive library of publicly-available transcription factor ChIP-sequencing and global chromatin accessibility profiles to infer transcriptional regulators. According to Lisa, MYC is in the top 2% of transcription factors predicted to influence upregulated genes in the PoWeR-trained transcriptome of aged mice (eight of 536 transcription factors) (Fig. 2B) and in the top 5% for downregulated genes (19 of 536) (see Supporting information, Table S4). MYC was the highest ranked among Yamanaka factors. MYC as a primary transcription factor controlling exercise adaptation in muscle is consistent with human biopsy data (Popov et al., 2019). MYC transcript and protein localizes in myonuclei during developmental and hypertrophic muscle growth in muscle of young animals (Alway, 1997; Armstrong & Esser, 2005; Murach et al., 2022; Veal & Jackson, 1998). Myc was 60% higher after late-life exercise in the soleus of PoWeR vs. sedentary mice (24 h after the final exercise bout, P = 0.013, adjusted P = 0.14) (Fig. 2C). We therefore aimed to explore the specific effects of a pulse of MYC in muscle fibres on muscle gene expression and compare it to the effects of OKSM and exercise training.
Figure 2. Identification of MYC-controlled genes in soleus muscle that also associate with late-life exercise and OKSM induction
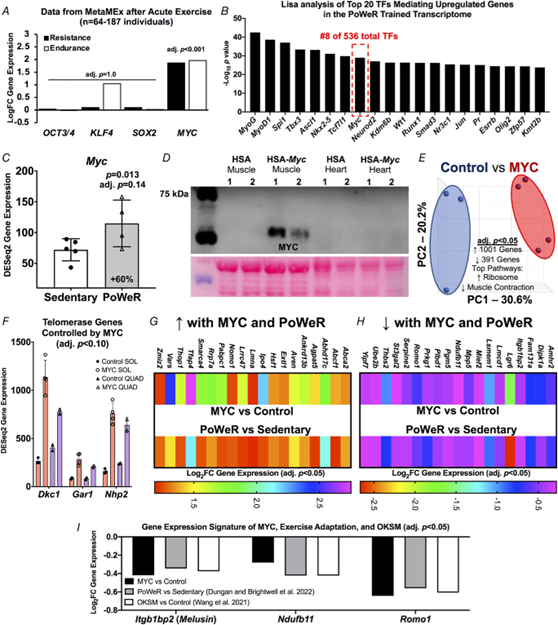
A, data from the MetaMEx human muscle and exercise meta-analysis tool from the Zierath laboratory (Pillon et al., 2020) showing the responsiveness of Oct3/4, Klf4, Sox2 and Myc to endurance and resistance exercise in humans. Input parameters: Male/Female, Young, Sedentary/Active/Athlete, Lean, Vastus Lateralis, Healthy, Immediate/0/3/4/5/6/8/18/24 hours. Adjusted p values are for each individual gene and condition. B, Lisa analysis of transcription factors (TFs) controlling upregulated genes in the PoWeR-trained transcriptome. C, Myc gene expression in the soleus after 8 weeks of PoWeR from 22 to 24 months in mice (Dungan, Brightwell et al., 2022). D, western blot showing MYC protein in skeletal muscle and heart 12 h after removal of overnight doxycycline in biological replicate MYC and control mice, with corresponding Ponceau S stain beneath. E, PCA plot of full soleus DESeq2 normalized gene count transcriptome dataset from MYC (n = 3) and control (n = 4) mice. F, telomerase complex genes controlled by MYC in the soleus (SOL) (n = 3 control, n = 4 MYC) and quadriceps (QUAD) (n = 2 control, n = 2 MYC) muscles. G, upregulated genes in the soleus common to MYC and late-life PoWeR. H, downregulated genes in the soleus common to MYC and late-life PoWeR. I, gene expression signature common to MYC, PoWeR and OKSM in the murine soleus. All data are reported as the mean ± SD.
Genetically controlled muscle-specific Myc induction recapitulates aspects of OKSM expression and exercise training adaptation
We generated a doxycycline-controlled muscle fibre-specific Myc expression model by crossing the HSA-rtTA mouse (Iwata et al., 2018) with the TetO-Myc mouse (Felsher & Bishop, 1999). This mouse is a tool for understanding the temporal control of MYC target genes in skeletal muscle fibres in vivo. Young adult mice had access to doxycycline in drinking water overnight resulting in Myc induction. Doxycycline was removed in the morning and soleus muscle was collected 12 h later (n = 3 HSA-rtTA littermate controls, n = 4 HSA-Myc). MYC protein was elevated at 12 h after doxycycline removal in muscle and global soleus gene expression was markedly altered at this time (Fig. 2D and E). Inputting the MYC-controlled genes (adjusted P < 0.05) from the soleus into Lisa (Qin et al., 2020), MYC is predicted as the highest ranked transcription factor regulating induced genes (data not shown); this confirms the veracity of Lisa and agrees with our previous findings in the plantaris muscle (Murach et al., 2022). MYC induced 1001 genes and repressed 391 genes in the soleus (adjusted P < 0.05) (see Supporting information, Table S5). Ribosomal genes were the most upregulated (adjusted P = 3.3 × 10−29, KEGG) and muscle contraction genes were downregulated (adjusted P = 0.0009, Reactome) (Fig. 2E). MYC can transiently repress cellular identity (Sullivan et al., 2022) and downregulation of muscle-enriched genes corresponds with OKSM partial reprogramming in myofibres (Fig. 1). We previously reported that a pulse of MYC downregulated Nr1d2 (Reverbβ) and upregulated Rpl3 in the plantaris muscle of the mice used here (Murach et al., 2022). These findings were corroborated in the soleus and we also report a significant repression of Rpl3l by MYC (adjusted P = 0.041) (see Supporting information, Table S5). This inverse pattern of Rpl3 (induction) and Rpl3l (repression) is indicative of a developmental- and growth-oriented gene expression program in skeletal muscle (Chaillou, 2019; Chaillou et al., 2016; Kao et al., 2021; Lin et al., 2022; Zhang, Trapp et al., 2022; Zhang, Englund et al., 2022; Zhang, Hong et al., 2022). OKSM expression vs. MYC induction revealed 39 upregulated genes and 31 downregulated genes in common. Ribosome biogenesis-related genes including Akt1, Mdn1, and Wdr3 were commonly upregulated and mitochondrial genes including Apoo, Ndufa1, Ndufb11, Pdcd5, and Romo1 were commonly downregulated (see Supporting information, Table S6). MYC recapitulated several aspects of OKSM-mediated partial reprogramming.
To further define the gene expression profile induced by MYC, we performed RNA-seq on the quadriceps muscles from a subset of HSA-Myc mice (n = 2 control and n = 2 Myc). The quadriceps are considerably larger than the soleus and are also involved in murine wheel training. Quadriceps were less affected by MYC and fewer genes were altered (40 upregulated and 26 downregulated at adjusted P < 0.10) (see Supporting information, Table S7) relative to the soleus and the plantaris, the latter of which we reported previously (Murach et al., 2022). Thus, the magnitude of MYC regulation may be muscle-specific, and could depend on muscle function, fibre type distribution and/or metabolic profile. Pathway analysis of genes altered by MYC induction in the quadriceps revealed downregulation of muscle contraction genes (e.g. various myosins and troponins) consistent with what is observed in the soleus (present study) and plantaris (Murach et al., 2022). MYC induced Dkc1, Gar1 and Nhp2 in the quadriceps which were also markedly upregulated by MYC in the soleus (Fig. 2F). These genes are members of the telomerase complex and associated with telomere maintenance (Gu et al., 2008; Vulliamy et al., 2008). Telomere shortening is associated with ageing (López-Otín et al., 2013). MYC is known to activate telomerase (Wang et al., 1998), which facilitates peripheral nervous system regeneration (Ma et al., 2019). Alterations to telomerase genes further implicate MYC as a potential age-mitigating reprogramming factor.
We next considered whether gene expression in the soleus after PoWeR in aged mice matched the MYC-controlled soleus transcriptome. Thirty-eight genes overlapped (19 up- and 19 downregulated, adjusted P < 0.05, Fig. 2G and H). Itgb1bp2 (Melusin), Ndufb11 and Romo1 were downregulated by MYC, PoWeR, and OKSM (Fig. 2I). Repression of the mitochondrial complex I accessory subunit Ndufb11 was common to all datasets including long-term human exercise training (adjusted P = 0.02) (Chapman et al., 2020) (Fig. 2E).
A pulse of MYC reshapes the global muscle methylome
Using the same soleus tissue from the RNA-seq experiment above, we performed RRBS to assess the global DNA methylome. At 12 h after overnight exposure to doxycycline in HSA-Myc mice, the muscle methylome underwent global CpG remodelling relative to controls (>1000 distinct differentially methylated CpGs across promoters, exons and introns at P < 0.0005 and minimum 10× coverage across all samples). MYC caused slight hypomethylation of promoter region CpGs and hypermethylation of CpGs in exons and introns relative to controls (Fig. 3A) (see Supporting information, Table S8). There was some agreement between promoter methylation status and gene expression mediated by MYC. Three genes (Cfap298, Marchf5 and Mettl1) had a hypomethylated promoter CpG and higher gene expression with MYC induction (Fig. 3B and C). Cfap298 controls cilia motility and polarization (Jaffe et al., 2016), but its role in skeletal muscle is not understood. Marchf5 (also called March5 or Mitol) is an E3 ubiquitin ligase required for mitochondrial fission and regulates mitochondrial dynamics (Nie et al., 2021). Mettl1 is an RNA methyltransferase, which is noteworthy because MYC was recently identified as a regulator of RNA methylation (Jansson et al., 2021). Perhaps global gene expression and methylation status would align more closely after a longer and/or more frequent period of MYC induction, if we assessed mRNA levels at a later timepoint after methylation analysis, or if we used a more comprehensive DNA methylation analysis. We previously published low-input RRBS from soleus myonuclei of late-life PoWeR mice vs. sedentary controls (Dungan, Brightwell et al., 2022). There was modest agreement in gene-level regulation of promoter and exon regions when comparing the soleus MYC methylome to the myonuclear methylome after PoWeR (see Supporting information, Table S9). A larger number of common genes with broadly intersecting regulation was apparent in introns (Fig. 3D and E). Collectively, these data suggest that MYC regulates DNA methylation in skeletal muscle and could be sufficient to induce epigenetic reprogramming, even after brief exposure.
Figure 3. A pulse of MYC alters the DNA methylome in skeletal muscle
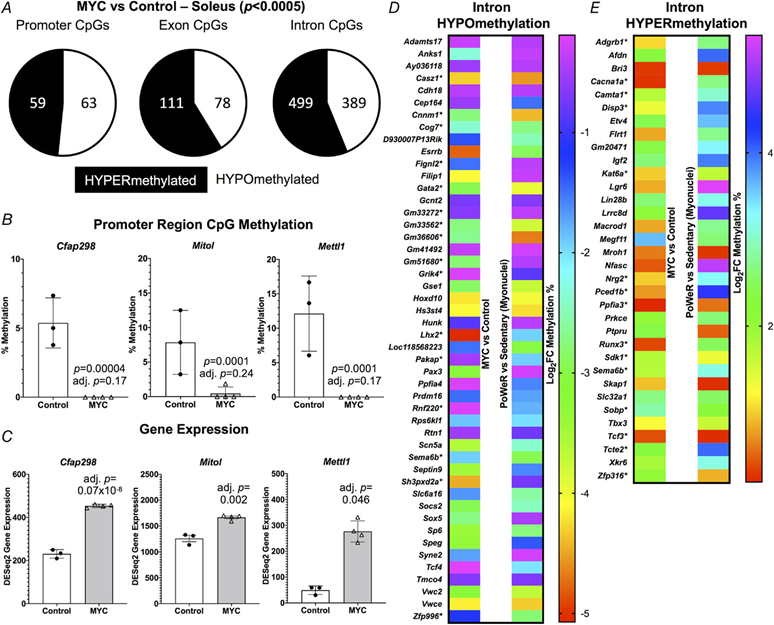
A, soleus muscle DNA methylation in promoter, exon and intron regions of HSA-Myc (n = 4) relative to control mice (n = 3), P < 0.0005 and 10× coverage per CpG site for MYC methylation data. Promoter CpG DNA methylation (B) and gene expression © of Cfap298, Mitol (Marchf5) and Mettl1 after a pulse of MYC, P < 0.0005 and 10× coverage per CpG site for MYC methylation data. Common gene-level intron hypo- (D) and hyper-methylation (E) with MYC induction in the soleus compared to soleus myonuclear DNA methylation after 8 weeks of late-life PoWeR (Dungan, Brightwell et al., 2022). *Multiple sites of regulation in one or both conditions, but only data from one site is shown. All data are reported as the mean ± SD.
Discussion
In the present study, we report:
- (1) a biological age-mitigating effect on the epigenetic landscape by late-life exercise-training in murine skeletal muscle;
- (2) a common gene expression signature of partial reprogramming by OKSM and exercise training in muscle of humans and aged mice; and
- (3) that Myc is an exercise-responsive factor that contributes to a rewired molecular profile at the transcriptome and methylome levels.
Lower muscle epigenetic age using updated and customized mDNAge clocks with exercise training corroborates and expands on our initial report in mice (Murach et al., 2021).
- Studies examining the muscle methylome after chronic exercise in aged humans also support our findings (Blocquiaux et al., 2022; Ruple et al., 2021; Sailani et al., 2019).
- Lower mDNAge in muscle appears to be a feature of the molecular environment with exercise adaptation that could have practical consequences for biological ageing and muscle performance.
- Indeed, emerging evidence suggests that blood mDNAge inversely associates with grip strength and frailty in ageing humans (Peterson et al., 2022; Verschoor et al., 2021) and is lower following a dietary/physical activity intervention in postmenopausal women (Fiorito et al., 2021).
- Exercise training and OKSM share substantial gene expression overlap.
- Given the known phenotypic and functional benefits of exercise adaptation in aged animals (Dungan, Brightwell et al., 2022), an altered transcriptome after brief OKSM-mediated epigenetic partial reprogramming in skeletal muscle could have functional consequences.
- Prior work shows that OKSM induction in muscle enhances regeneration after injury (Ocampo, Reddy, Martinez-Redondo et al., 2016; Wang et al., 2021), although more work is needed in the context of muscle mass, strength, quality, fatigue resistance and senescent cell abundance with ageing.
- Exploration into the interaction of epigenetic reprogramming via OKSM in combination with exercise training is also warranted.
Both OKSM and late-life exercise adaption in mice as well as chronic exercise training in humans results in lower levels of Abra/Stars in muscle.
- Exploratory proteomics recently found that low ABRA/STARS in human skeletal muscle is strongly associated with high physical activity throughout the lifespan (Ubaida-Mohien et al., 2019).
- ABRA/STARS has numerous functions in muscle (Lamon et al., 2014), but knockdown in myotubes enhances insulin signalling and sensitivity (Jin et al., 2011).
- Because insulin resistance associates with ageing (Barzilai & Ferrucci, 2012; Fink et al., 1983), lower ABRA/STARS in muscle could be beneficial for improving muscle health throughout the lifespan.
- Itgb1bp2 (Melusin), Ndufb11 and Romo1 were downregulated by OKSM expression, late-life exercise training and a pulse of MYC in the muscle of mice.
- Melusin is specific to striated muscle and increases during regeneration (Brancaccio et al., 1999).
- Conversely, Melusin decreases during atrophy (Vitadello et al., 2020), but its role in exercise adaptation is unclear.
- ROMO1 generates reactive oxygen species (Chung et al., 2006) and induces senescence (Chung et al., 2008).
- Downregulation of Romo1 may be beneficial in ageing muscle where senescent cells can manifest and impair adaptation (Dungan, Figueiredo et al., 2022; Dungan, Murach et al., 2022; Zhang, Englund et al., 2022; Zhang, Hong et al., 2022).
- ROMO1 is also a negative feedback regulator of MYC (Lee et al., 2011), and so its downregulation by MYC induction seems intuitive.
- Repressed gene expression of the complex I accessory subunit Ndufb11 with OKSM, Myc and exercise training in mice is conserved with chronic exercise in humans.
- Reduced complex I activity prevents reactive oxygen species production, which enhances cellular fitness and longevity (Rodríguez-Nuevo et al., 2022).
- Low abundance of specific complex I components including NDUFB11 is a characteristic of tissue from long-lived animals (Miwa et al., 2014; Sahm et al., 2018).
- Lower Nudfb11 with chronic exercise, probably mediated by MYC, could be part of a larger adaptive response that defends trained skeletal muscle against oxidative damage (Criswell et al., 1993; Parise, Brose et al., 2005; Parise, Phillips et al., 2005; Powers et al., 1999; Radak et al., 1999; Smuder et al., 2011).
- On balance, studies with ageing and exercise in muscle of humans report a disconnect between mitochondrial gene and protein abundance (Robinson et al., 2017; Tumasian III et al., 2021).
- Our results should be interpreted with this in mind.
- Future Ndufb11 gain- and loss-of-function experiments in muscle may be warranted to elucidate its potential role in combatting an aged muscle phenotype.
Myc gene and protein expression is induced in the muscle of young adult humans … in response to loading.
Myc gene and protein expression is induced in the muscle of young adult humans (Broholm et al., 2011; Brook et al., 2016; Figueiredo et al., 2016; Figueiredo et al., 2021; Popov et al., 2019; Stec et al., 2016; Townsend et al., 2016; Trenerry et al., 2007) and rodents (Armstrong & Esser, 2005; Chen et al., 2002; Goodman et al., 2015; Kirby et al., 2016; Lai et al., 2010; Murach et al., 2022; von Walden et al., 2012; West et al., 2016; Whitelaw & Hesketh, 1992) in response to loading.
- MYC not only participates in epigenetic reprogramming in concert with Oct3/4, Klf4 and Sox2 (Takahashi & Yamanaka, 2006), but can also facilitate epigenome remodelling on its own (Brenner et al., 2005; Gartel, 2006; Lin et al., 2009; Nakagawa et al., 2010; Pang et al., 2018; Poole et al., 2017; Poole et al., 2019).
- Consistent with work in other cell types, there was global remodelling of the muscle DNA methylome following MYC induction. We speculate this could be a result of interactions of MYC with DNA methyltransferases and ten eleven translocases (Brenner et al., 2005; Pang et al., 2018; Poole et al., 2019).
- It is striking that <24 h of MYC induction can change muscle DNA methylation status, but a single bout of exercise also remodels DNA methylation in human muscle tissue (Barres et al., 2012; Maasar et al., 2021; Seaborne et al., 2018).
- The mechanisms of methylation regulation in muscle by exercise are unclear (Small et al., 2021; Villivalam et al., 2021), but MYC could be a central factor.
- Recent evidence suggests that overexpression of MYC in aged oligodendrocyte progenitors is sufficient to restore regenerative remyelination in vivo (Neumann et al., 2021); this is consistent with the role of MYC in regulating in vivo sensory nerve regeneration (Ma et al., 2019).
- Short-term MYC expression recapitulates numerous aspects of myonuclear gene expression at the onset of rapid overload-mediated muscle hypertrophy (Murach et al., 2022). T
- wo weeks of MYC overexpression also mimics the protein synthesis response to exercise-like high-intensity muscle contractions (Mori et al., 2021).
- Because MYC-controlled gene expression in muscle fibres overlaps with OKSM partial reprogramming and the exercise training-mediated transcriptomes of aged mice, we propose that pulses of Myc could serve to enhance muscle function.
A 60% induction of Myc in the soleus after late-life PoWeR is intriguing because whole-organism MYC knockdown may enhance longevity and healthspan in mice (Hofmann et al., 2015).
By contrast, MYC induction in specific cell populations can attenuate cellular ageing and restore regenerative potential (Neumann et al., 2021). Myc gene and protein in muscle tissue may increase in rodents (Hofmann et al., 2014; Mobley et al., 2017) but not humans during ageing (Drummond et al., 2011; Stec et al., 2015).
- The behaviour of MYC in muscle with ageing is probably complex and could be differentially affected in mononuclear proliferative cells vs. multinuclear post-mitotic muscle fibres within muscle tissue.
- Single cell RNA-sequencing in muscle revealed Myc enrichment specifically in fibro/adipogenic progenitors, satellite cells, and tenocytes but not myonuclei of aged mice (Zhang, Hong et al., 2022).
- Elevated Myc in satellite cells of aged mice has been previously described (Price et al., 2014).
- Cell type-specific upregulation of Myc may explain differing results at the tissue level.
- Aspects of the gene expression network of Myc, which is operative after muscle contraction when young (Murach et al., 2022; Popov et al., 2019), may be alternatively regulated after resistance training in human muscle when aged (Phillips et al., 2013).
- MYC gene expression in vastus lateralis muscle samples 4 h after a bout of resistance exercise in ∼80-year-old men and women is blunted (logFC = 0.64 and −0.54, respectively, adjusted P > 0.90) relative to ∼24-year-old men and women (logFC = 1.84 and 1.73, respectively, adjusted P < 0.05, data extracted from MetaMEx) (Pillon et al., 2020; Raue et al., 2012).
- Blunted MYC gene and protein responsiveness to acute resistance exercise in muscle of younger, albeit still aged humans (∼70 years) has also been observed (Brook et al., 2016; Rivas et al., 2014).
- Perhaps an attenuated MYC response in muscle fibres of very old humans and animals (Alway, 1997) explains diminished adaptability to exercise, specifically in glycolytic type 2 muscle fibres (Grosicki et al., 2022; Raue et al., 2009; Slivka et al., 2008).
- We speculate that higher Myc levels in the oxidative soleus muscle after high-volume PoWeR in aged mice is favourable and could contribute to the notable cellular adaptations observed previously (Dungan, Brightwell et al., 2022).
Tighter control over the duration of OKSM and Myc dosing, the timing of sampling after exercise and genetically-induced epigenetic partial reprogramming, the mode of exercise and training status, the potential effects of biological sex, and several other factors …
… may identify a larger gene expression signature of exercise-associated reprogramming-linked genes in muscle.
Importantly, understanding whether epigenetic reprogramming, specifically by MYC, induces functional consequences in skeletal muscle is essential.
- Such insight will help drive healthspan-extending therapeutic efforts forward and advance the ageing field beyond biomarker discovery.
- Because DNA demethylation is required for cellular reprogramming (Simonsson & Gurdon, 2004), the precise mechanisms by which MYC reshapes the methylome and global epigenome in muscle also deserves further exploration.
- Ultimately, the mechanisms of muscle molecular reprogramming required to fully recapitulate a youthful phenotype remain elusive, but our data provide a roadmap for further examination of how exercise training combats aspects of ageing.
- Our data also implicate MYC as an exercise-induced reprogramming factor in skeletal muscle.
DEEP DIVE

Can exercise reverse muscle aging? Yes, and this is how
Medical News Today
James Kingsland , Fact checked by Hannah Flynn
January 24, 2023 —
Research shows that people who exercise regularly not only strengthen their muscles but also improve their overall health, regardless of how late in life they start.
- For example, recent studies have found that exercise reduces the risk of cardiovascular disease,
- as well as Alzheimer’s and Parkinson’s in older people.
- Conversely, reductions in muscle mass and strength are associated with lower quality of life and higher mortality from all causes.
As a result of its proven ability to prevent and treat several chronic diseases at low cost, doctors have called exercise a drug-free “polypill” that can benefit nearly everyone.
“Exercise is the most powerful drug we have,” says Dr. Kevin Murach, assistant professor at the Exercise Science Research Center, University of Arkansas, Fayetteville, AR.
He believes that exercise should be considered a health-enhancing, potentially life-extending treatment, alongside medications and a healthy diet.
“Exercise is the most powerful drug we have…
exercise should be considered a health-enhancing, potentially life-extending treatment, alongside medications and a healthy diet.

Scientists hope that a better understanding of how exercise rejuvenates aged muscle at a molecular level will provide clues for future anti-aging therapies.
Exercise may turn back the clock in muscle fibers by promoting the “epigenetic reprogramming” of chromosomes in the cells’ nuclei.
Epigenetics refers to how chemical changes affect the activity or “expression” of genes.
For example, proteins called transcription factors can dial up the expression of particular genes when they bind to specific DNA sequences.
In 2012, Dr. Shinya Yamanaka shared the Nobel Prize for Medicine for his discovery that four transcription factors can revert specialized, mature cells to more youthful, flexible cells called pluripotent stem cells.
The four factors are called Oct3/4, Klf4, Sox2, and Myc, or OKSM for short.
In 2012, Dr. Shinya Yamanaka shared the Nobel Prize for Medicine for his discovery that four transcription factors can revert specialized, mature cells to more youthful, flexible cells called pluripotent stem cells.
The four factors are called Oct3/4, Klf4, Sox2, and Myc, or OKSM for short.

In a new study whose results appear in The Journal of Physiology, Dr. Murach and his colleagues compared the effects of OKSM factors on gene expression in the muscle fibers of mice …
…that had access to an exercise wheel, and mice that had no access.
In addition, they compared the effects of OKSM factors on muscle with the effects of a single transcription factor, Myc. Scientists have found that exercise induces the expression of Myc to a greater extent than the other three factors.
The researchers also investigated how exercise alone affected gene expression in muscle fibers from both mice and humans.
The mice were 22 months old, which is equivalent to a human age of around 73 years.
Mice in the exercise group were free to run on an unweighted wheel for the first week, then, over the next 8 weeks, the scientists made the wheel progressively heavier by attaching magnetic weights to it.
The results suggest that exercise reprograms muscle fibers to a more youthful state through increased expression of the genes that make Yamanaka factors, in particular Myc.
The results suggest that exercise reprograms muscle fibers to a more youthful state through increased expression of the genes that make Yamanaka factors, in particular Myc.
Dr. Murach suggests that the findings may one day lead to the development of drugs that supercharge the exercise response of muscles in people who are confined to bed, or the muscles of astronauts in zero gravity.
… the findings may one day lead to the development of drugs that supercharge the exercise response of muscles in people who are confined to bed, or the muscles of astronauts in zero gravity.

But he dismisses the idea of a pill that boosts the expression of Myc ever replacing the need to exercise.
For one thing, exercise has beneficial effects throughout the body, not just in muscle.
In addition, Myc has been linked to cancer, so there are inherent risks in artificially boosting its expression.
In addition, Myc has been linked to cancer, so there are inherent risks in artificially boosting its expression.

In their paper, the researchers also note that drugs that are gaining a popular reputation as “life-extending” may actually block some of the beneficial effects of exercise on muscle.
Dr. Murach told Medical News Today:
“Evidence suggests that ‘life-extending’ drugs such as metformin and rapamycin interfere with the positive benefits of exercise specifically in skeletal muscle.”
He said it was “not outside the realm of possibility” that the drugs could disrupt the epigenetic reprogramming of muscle that happens with exercise.

MNT asked exercise physiologists to recommend the best type of exercise for older people.
“For individuals over 70 I would highly recommend low-impact, full-body workouts with a focus on lower body and core,” advised John C. Loges, an exercise physiologist at eVOLV Strong.
“For individuals over 70 I would highly recommend low-impact, full-body workouts with a focus on lower body and core,” …
“Resistance training is not only suitable but highly recommended for those in their 70s and beyond,” he said.
“The key is starting slow and progressing slowly with consistency,” he added.
“[W]alking is an activity that I recommend, along with resistance and mobility training,” advised Melissa Hendrix Wogahn, an exercise physiologist at Joy of Active Living who offers fitness and health education for older adults.
She recommended strength training at least two days a week and mobility training, including stretching, every day.
“[W]alking is an activity that I recommend, along with resistance and mobility training, …
… strength training at least two days a week and mobility training, including stretching, every day.
“In terms of frequency, an older adult can walk every day, assuming they have no contraindications,” she added.
The authors of the new study acknowledge that it had some limitations.
For example, the type of exercise, training status, biological sex, and several other factors may affect gene expression changes associated with exercise.
In addition, they emphasize the importance of investigating the functional consequences of epigenetic reprogramming in skeletal muscle.
Originally published at https://www.medicalnewstoday.com on January 24, 2023.
ORIGINAL PUBLICATION

A molecular signature defining exercise adaptation with ageing and in vivo partial reprogramming in skeletal muscle [excerpt version]
The Journal of Phisiology (The Physiological Society)
Ronald G. Jones III, Andrea Dimet-Wiley, Amin Haghani, Francielly Morena da Silva, Camille R. Brightwell, Seongkyun Lim, Sabin Khadgi, Yuan Wen, Cory M. Dungan, Robert T. Brooke, Nicholas P. Greene, Charlotte A. Peterson, John J. McCarthy, Steve Horvath, Stanley J. Watowich, Christopher S. Fry, Kevin A. Murach … See fewer authors
19 December 2022
Abstract
- Exercise promotes functional improvements in aged tissues, but the extent to which it simulates partial molecular reprogramming is unknown.
- Using transcriptome profiling from (1) a skeletal muscle-specific in vivo Oct3/4, Klf4, Sox2 and Myc (OKSM) reprogramming-factor expression murine model; (2) an in vivo inducible muscle-specific Myc induction murine model; (3) a translatable high-volume hypertrophic exercise training approach in aged mice; and (4) human exercise muscle biopsies, we collectively defined exercise-induced genes that are common to partial reprogramming.
- Late-life exercise training lowered murine DNA methylation age according to several contemporary muscle-specific clocks.
- A comparison of the murine soleus transcriptome after late-life exercise training to the soleus transcriptome after OKSM induction revealed an overlapping signature that included higher JunB and Sun1.
- Also, within this signature, downregulation of specific mitochondrial and muscle-enriched genes was conserved in skeletal muscle of long-term exercise-trained humans; among these was muscle-specific Abra/Stars.
- Myc is the OKSM factor most induced by exercise in muscle and was elevated following exercise training in aged mice.
- A pulse of MYC rewired the global soleus muscle methylome, and the transcriptome after a MYC pulse partially recapitulated OKSM induction.
- A common signature also emerged in the murine MYC-controlled and exercise adaptation transcriptomes, including lower muscle-specific Melusin and reactive oxygen species-associated Romo1.
- With Myc, OKSM and exercise training in mice, as well habitual exercise in humans, the complex I accessory subunit Ndufb11 was lower; low Ndufb11 is linked to longevity in rodents.
- Collectively, exercise shares similarities with genetic in vivo partial reprogramming.
Key points
- Advances in the last decade related to cellular epigenetic reprogramming (e.g. DNA methylome remodelling) toward a pluripotent state via the Yamanaka transcription factors Oct3/4, Klf4, Sox2 and Myc (OKSM) provide a window into potential mechanisms for combatting the deleterious effects of cellular ageing.
- Using global gene expression analysis, we compared the effects of in vivo OKSM-mediated partial reprogramming in skeletal muscle fibres of mice to the effects of late-life murine exercise training in muscle.
- Myc is the Yamanaka factor most induced by exercise in skeletal muscle, and so we compared the MYC-controlled transcriptome in muscle to Yamanaka factor-mediated and exercise adaptation mRNA landscapes in mice and humans.
- A single pulse of MYC is sufficient to remodel the muscle methylome.
- We identify partial reprogramming-associated genes that are innately altered by exercise training and conserved in humans, and propose that MYC contributes to some of these responses.

METHODS & OTHER SECTIONS
See the original publication (this is an excerpt version_

RESULTS
Late-life exercise training associates with lower skeletal muscle mDNAge using contemporary muscle-specific epigenetic clocks
To corroborate our previously-described exercise training-mediated shift in methylation age toward a younger status in muscle, female C57BL/6N mice (n = 10) completed PoWeR from 22 to 24 months. Approximately 22-month-old mice are estimated to be equivalent to ∼73-year-old humans (Dutta & Sengupta, 2016). Age-matched untrained mice were sedentary controls (n = 9). Muscle methylation was quantified with the Horvath Mammalian Methylation Chip that focuses on conserved cytosines (Arneson et al., 2022) and analysed with contemporary murine mDNAge clocks (Mozhui et al., 2022). After PoWeR, skeletal muscle mDNAge was lower relative to sedentary using the Horvath Muscle Clock (−5.7 weeks, P = 0.04) and Developmental Muscle Clock (−7.1 weeks, P = 0.02) (Fig. 1A). The magnitude of epigenetic age mitigation by late-life exercise in muscle was similar to our previous report using an independent cohort of mice and a different mDNAge analysis (8 weeks, n = 5 per group, P = 0.07) (Murach et al., 2021). Running volume was slightly lower in the present study (4.3 km day–1 vs. 6.4 km day–1). Consistently lower mDNAge with exercise motivated us to compare transcriptome profiles of a partial reprogramming stimulus (OKSM expression) vs. exercise adaptation in muscle to identify a common gene expression signature.
Figure 1. Comparison of Oct3/4, Klf4, Sox2 and Myc (OKSM) expression to late-life progressive weighted wheel running (PoWeR) in mouse muscle

A, DNA methylation age (mDNAge) derived from clocks developed by the Horvath laboratory applied to late-life PoWeR (n = 10) and sedentary control (n = 9) mouse gastrocnemius muscle. B, PCA plots showing DESeq2-normalized gene count transcriptome data from OKSM expression (n = 2) vs. control (n = 2) in mouse soleus muscle fibres (Wang et al., 2021) and the late-life PoWeR (n = 4) vs. the sedentary (n = 5) transcriptome in soleus muscle (Dungan, Brightwell et al., 2022). C, chord diagram showing significantly downregulated genes common across OKSM and late-life PoWeR in the soleus. D, upregulated genes common across OKSM and late-life PoWeR in the soleus. E, downregulated genes common across OKSM, late-life PoWeR and long-term exercise training in humans (Chapman et al.) All data are reported as the mean ± SD.
The transcriptome resulting from OKSM expression in muscle fibres overlaps with the late-life exercise adaptation transcriptome in mouse muscle
RNA-seq data from the soleus muscle of adult mice after 2.5 days of genetic OKSM expression (Wang et al., 2021) was compared to RNA-sequencing data from the soleus of late-life PoWeR mice (Dungan, Brightwell et al., 2022). The soleus adapts more substantially to PoWeR than other hindlimb muscles in young and old mice (Englund et al., 2020; Murach et al., 2020), with similar gene expression patterns regardless of age (Dungan, Brightwell et al., 2022). The soleus also contains similar proportions of the primary myosin fibre types found in human vastus lateralis (Dungan, Brightwell et al., 2022; Murach et al., 2019, 2020), further justifying our focus on this muscle.
OKSM induction and PoWeR revealed stark effects on gene expression for each intervention (Fig. 1B). With OKSM, 412 genes were upregulated and 523 genes were downregulated (adjusted P < 0.05) (see Supporting information, Table S1). Pathway analysis of OKSM induction revealed repression of genes associated with oxidative phosphorylation, in agreement with the analysis from the Belmonte laboratory (Wang et al., 2021). We compared the list of genes downregulated by OKSM with those downregulated by PoWeR (377 genes, adjusted P < 0.05) (see Supporting information, Table S2). Approximately one-quarter of downregulated genes overlapped (87 genes) (Fig. 1C). Pathway analysis of shared downregulated genes revealed oxidative phosphorylation (KEGG, adjusted P = 9.77 × 10−54), the citric acid cycle (Reactome, adjusted P = 8.31 × 10−56) and electron transport chain (Wikipathways, adjusted P = 2.08 × 10−44) as the top pathways (Fig. 1C). The muscle-enriched genes fast skeletal muscle troponin (Tnni2) (Sheng & Jin, 2016) and striated muscle activator of Rho signalling (Stars, or Abra) (Arai et al., 2002; Wallace et al., 2012) were also lower with OKSM and PoWeR; this suggested a repression of muscle cellular identity, consistent with partial reprogramming effects. Seventeen upregulated genes were common between OKSM and PoWeR datasets (Fig. 1D), including the potent muscle mass-regulator JunB (Raffaello et al., 2010) and myonuclear anchor Sun1 (Lei et al., 2009). Perhaps alteration to Sun1 could be related to myonuclear shape changes that occur with exercise training in rodents (Battey et al., 2023; Murach et al., 2020; Rader & Baker, 2022). Collectively, our analysis unearthed a canonical skeletal muscle gene expression signature common between OKSM and exercise adaptation.
Gene expression alterations with OKSM and exercise adaptation in mouse muscle are conserved in human muscle after prolonged exercise training
To validate our murine findings in humans we queried an RNA-sequencing dataset from muscle biopsy samples of women with at least 15 years of mixed-modality endurance training history (n = 9, ∼42 years old) (Chapman et al., 2020). Comparing our murine PoWeR data to long-term training in humans seemed appropriate as 8 weeks of high-volume PoWeR corresponds to ∼10% of the mouse lifespan and could thus be characterized as prolonged training. Several mitochondrial-related genes downregulated by OKSM and PoWeR were significantly lower in muscle of long-term endurance-trained athletes relative to sedentary age-matched controls (COX6A2, adjusted P = 0.046; NDUFB11, adjusted P = 0.017; NDUFS7, adjusted P = 0.041; TOMM7, adjusted P = 0.028). Lower levels of mitochondrial genes with endurance training appears to be counterintuitive. To further explore this observation, we compared genes that were altered in female endurance trained muscle to the human MitoCarta 3.0 database (Rath et al., 2021). Although 100 genes that were higher with training overlapped with MitoCarta, 52 genes that were lower also overlapped; these included NDUFs such as NDUFAF3 and NDUFS7, as well as COX6A1 and COX6A2 (see Supporting information, Table S3). Thus, endurance training adaptations in skeletal muscle are not characterized by unanimously elevated mitochondrial gene levels. In addition to mitochondrial genes, muscle-enriched TNNI2 and ABRA (STARS) were lower in exercise-trained human muscle (adjusted P = 0.002 and 0.003, respectively), consistent with OKSM expression and late-life PoWeR in mice (Fig. 1E).
Myc is an exercise-responsive transcription factor in muscle that influences the gene expression landscape with PoWeR in aged mice
The OKSM genetic mouse model was designed to induce all four Yamanaka factors, but Klf4 gene expression was not increased significantly in the soleus in the OKSM experiment (Wang et al., 2021); we corroborate this in our analysis (1.05 Fold change (FC) OKSM vs. Sedentary, adjusted P = 0.95). All four factors can contribute to muscle adaptation via a change in activity independent of expression levels, but partial reprogramming was probably most attributable to Oct3/4, Sox2 and Myc. Of OKSM, only MYC is significantly induced by a bout of exercise in skeletal muscle from healthy humans according to a transcriptome meta-analysis (n = 64–187 subjects, acute resistance and endurance exercise) (Pillon et al., 2020) (Fig. 2A). It is worth noting that the KLF4 response is quite variable but may be induced in the first few hours after endurance exercise. Using up- and downregulated differentially expressed gene lists from our PoWeR RNA-seq data in the soleus, we performed Lisa analysis for insight on the potential contribution of MYC in the control of gene expression (Murach et al., 2022; Qin et al., 2020). Lisa incorporates transcriptome input data with an extensive library of publicly-available transcription factor ChIP-sequencing and global chromatin accessibility profiles to infer transcriptional regulators. According to Lisa, MYC is in the top 2% of transcription factors predicted to influence upregulated genes in the PoWeR-trained transcriptome of aged mice (eight of 536 transcription factors) (Fig. 2B) and in the top 5% for downregulated genes (19 of 536) (see Supporting information, Table S4). MYC was the highest ranked among Yamanaka factors. MYC as a primary transcription factor controlling exercise adaptation in muscle is consistent with human biopsy data (Popov et al., 2019). MYC transcript and protein localizes in myonuclei during developmental and hypertrophic muscle growth in muscle of young animals (Alway, 1997; Armstrong & Esser, 2005; Murach et al., 2022; Veal & Jackson, 1998). Myc was 60% higher after late-life exercise in the soleus of PoWeR vs. sedentary mice (24 h after the final exercise bout, P = 0.013, adjusted P = 0.14) (Fig. 2C). We therefore aimed to explore the specific effects of a pulse of MYC in muscle fibres on muscle gene expression and compare it to the effects of OKSM and exercise training.
Figure 2. Identification of MYC-controlled genes in soleus muscle that also associate with late-life exercise and OKSM induction

A, data from the MetaMEx human muscle and exercise meta-analysis tool from the Zierath laboratory (Pillon et al., 2020) showing the responsiveness of Oct3/4, Klf4, Sox2 and Myc to endurance and resistance exercise in humans. Input parameters: Male/Female, Young, Sedentary/Active/Athlete, Lean, Vastus Lateralis, Healthy, Immediate/0/3/4/5/6/8/18/24 hours. Adjusted p values are for each individual gene and condition. B, Lisa analysis of transcription factors (TFs) controlling upregulated genes in the PoWeR-trained transcriptome. C, Myc gene expression in the soleus after 8 weeks of PoWeR from 22 to 24 months in mice (Dungan, Brightwell et al., 2022). D, western blot showing MYC protein in skeletal muscle and heart 12 h after removal of overnight doxycycline in biological replicate MYC and control mice, with corresponding Ponceau S stain beneath. E, PCA plot of full soleus DESeq2 normalized gene count transcriptome dataset from MYC (n = 3) and control (n = 4) mice. F, telomerase complex genes controlled by MYC in the soleus (SOL) (n = 3 control, n = 4 MYC) and quadriceps (QUAD) (n = 2 control, n = 2 MYC) muscles. G, upregulated genes in the soleus common to MYC and late-life PoWeR. H, downregulated genes in the soleus common to MYC and late-life PoWeR. I, gene expression signature common to MYC, PoWeR and OKSM in the murine soleus. All data are reported as the mean ± SD.
Genetically controlled muscle-specific Myc induction recapitulates aspects of OKSM expression and exercise training adaptation
We generated a doxycycline-controlled muscle fibre-specific Myc expression model by crossing the HSA-rtTA mouse (Iwata et al., 2018) with the TetO-Myc mouse (Felsher & Bishop, 1999). This mouse is a tool for understanding the temporal control of MYC target genes in skeletal muscle fibres in vivo. Young adult mice had access to doxycycline in drinking water overnight resulting in Myc induction. Doxycycline was removed in the morning and soleus muscle was collected 12 h later (n = 3 HSA-rtTA littermate controls, n = 4 HSA-Myc). MYC protein was elevated at 12 h after doxycycline removal in muscle and global soleus gene expression was markedly altered at this time (Fig. 2D and E). Inputting the MYC-controlled genes (adjusted P < 0.05) from the soleus into Lisa (Qin et al., 2020), MYC is predicted as the highest ranked transcription factor regulating induced genes (data not shown); this confirms the veracity of Lisa and agrees with our previous findings in the plantaris muscle (Murach et al., 2022). MYC induced 1001 genes and repressed 391 genes in the soleus (adjusted P < 0.05) (see Supporting information, Table S5). Ribosomal genes were the most upregulated (adjusted P = 3.3 × 10−29, KEGG) and muscle contraction genes were downregulated (adjusted P = 0.0009, Reactome) (Fig. 2E). MYC can transiently repress cellular identity (Sullivan et al., 2022) and downregulation of muscle-enriched genes corresponds with OKSM partial reprogramming in myofibres (Fig. 1). We previously reported that a pulse of MYC downregulated Nr1d2 (Reverbβ) and upregulated Rpl3 in the plantaris muscle of the mice used here (Murach et al., 2022). These findings were corroborated in the soleus and we also report a significant repression of Rpl3l by MYC (adjusted P = 0.041) (see Supporting information, Table S5). This inverse pattern of Rpl3 (induction) and Rpl3l (repression) is indicative of a developmental- and growth-oriented gene expression program in skeletal muscle (Chaillou, 2019; Chaillou et al., 2016; Kao et al., 2021; Lin et al., 2022; Zhang, Trapp et al., 2022; Zhang, Englund et al., 2022; Zhang, Hong et al., 2022). OKSM expression vs. MYC induction revealed 39 upregulated genes and 31 downregulated genes in common. Ribosome biogenesis-related genes including Akt1, Mdn1, and Wdr3 were commonly upregulated and mitochondrial genes including Apoo, Ndufa1, Ndufb11, Pdcd5, and Romo1 were commonly downregulated (see Supporting information, Table S6). MYC recapitulated several aspects of OKSM-mediated partial reprogramming.
To further define the gene expression profile induced by MYC, we performed RNA-seq on the quadriceps muscles from a subset of HSA-Myc mice (n = 2 control and n = 2 Myc). The quadriceps are considerably larger than the soleus and are also involved in murine wheel training. Quadriceps were less affected by MYC and fewer genes were altered (40 upregulated and 26 downregulated at adjusted P < 0.10) (see Supporting information, Table S7) relative to the soleus and the plantaris, the latter of which we reported previously (Murach et al., 2022). Thus, the magnitude of MYC regulation may be muscle-specific, and could depend on muscle function, fibre type distribution and/or metabolic profile. Pathway analysis of genes altered by MYC induction in the quadriceps revealed downregulation of muscle contraction genes (e.g. various myosins and troponins) consistent with what is observed in the soleus (present study) and plantaris (Murach et al., 2022). MYC induced Dkc1, Gar1 and Nhp2 in the quadriceps which were also markedly upregulated by MYC in the soleus (Fig. 2F). These genes are members of the telomerase complex and associated with telomere maintenance (Gu et al., 2008; Vulliamy et al., 2008). Telomere shortening is associated with ageing (López-Otín et al., 2013). MYC is known to activate telomerase (Wang et al., 1998), which facilitates peripheral nervous system regeneration (Ma et al., 2019). Alterations to telomerase genes further implicate MYC as a potential age-mitigating reprogramming factor.
We next considered whether gene expression in the soleus after PoWeR in aged mice matched the MYC-controlled soleus transcriptome. Thirty-eight genes overlapped (19 up- and 19 downregulated, adjusted P < 0.05, Fig. 2G and H). Itgb1bp2 (Melusin), Ndufb11 and Romo1 were downregulated by MYC, PoWeR, and OKSM (Fig. 2I). Repression of the mitochondrial complex I accessory subunit Ndufb11 was common to all datasets including long-term human exercise training (adjusted P = 0.02) (Chapman et al., 2020) (Fig. 2E).
A pulse of MYC reshapes the global muscle methylome
Using the same soleus tissue from the RNA-seq experiment above, we performed RRBS to assess the global DNA methylome. At 12 h after overnight exposure to doxycycline in HSA-Myc mice, the muscle methylome underwent global CpG remodelling relative to controls (>1000 distinct differentially methylated CpGs across promoters, exons and introns at P < 0.0005 and minimum 10× coverage across all samples). MYC caused slight hypomethylation of promoter region CpGs and hypermethylation of CpGs in exons and introns relative to controls (Fig. 3A) (see Supporting information, Table S8). There was some agreement between promoter methylation status and gene expression mediated by MYC. Three genes (Cfap298, Marchf5 and Mettl1) had a hypomethylated promoter CpG and higher gene expression with MYC induction (Fig. 3B and C). Cfap298 controls cilia motility and polarization (Jaffe et al., 2016), but its role in skeletal muscle is not understood. Marchf5 (also called March5 or Mitol) is an E3 ubiquitin ligase required for mitochondrial fission and regulates mitochondrial dynamics (Nie et al., 2021). Mettl1 is an RNA methyltransferase, which is noteworthy because MYC was recently identified as a regulator of RNA methylation (Jansson et al., 2021). Perhaps global gene expression and methylation status would align more closely after a longer and/or more frequent period of MYC induction, if we assessed mRNA levels at a later timepoint after methylation analysis, or if we used a more comprehensive DNA methylation analysis. We previously published low-input RRBS from soleus myonuclei of late-life PoWeR mice vs. sedentary controls (Dungan, Brightwell et al., 2022). There was modest agreement in gene-level regulation of promoter and exon regions when comparing the soleus MYC methylome to the myonuclear methylome after PoWeR (see Supporting information, Table S9). A larger number of common genes with broadly intersecting regulation was apparent in introns (Fig. 3D and E). Collectively, these data suggest that MYC regulates DNA methylation in skeletal muscle and could be sufficient to induce epigenetic reprogramming, even after brief exposure.
Figure 3. A pulse of MYC alters the DNA methylome in skeletal muscle

A, soleus muscle DNA methylation in promoter, exon and intron regions of HSA-Myc (n = 4) relative to control mice (n = 3), P < 0.0005 and 10× coverage per CpG site for MYC methylation data. Promoter CpG DNA methylation (B) and gene expression © of Cfap298, Mitol (Marchf5) and Mettl1 after a pulse of MYC, P < 0.0005 and 10× coverage per CpG site for MYC methylation data. Common gene-level intron hypo- (D) and hyper-methylation (E) with MYC induction in the soleus compared to soleus myonuclear DNA methylation after 8 weeks of late-life PoWeR (Dungan, Brightwell et al., 2022). *Multiple sites of regulation in one or both conditions, but only data from one site is shown. All data are reported as the mean ± SD.
Discussion
In the present study, we report: (1) a biological age-mitigating effect on the epigenetic landscape by late-life exercise-training in murine skeletal muscle; (2) a common gene expression signature of partial reprogramming by OKSM and exercise training in muscle of humans and aged mice; and (3) that Myc is an exercise-responsive factor that contributes to a rewired molecular profile at the transcriptome and methylome levels.
Lower muscle epigenetic age using updated and customized mDNAge clocks with exercise training corroborates and expands on our initial report in mice (Murach et al., 2021). Studies examining the muscle methylome after chronic exercise in aged humans also support our findings (Blocquiaux et al., 2022; Ruple et al., 2021; Sailani et al., 2019). Lower mDNAge in muscle appears to be a feature of the molecular environment with exercise adaptation that could have practical consequences for biological ageing and muscle performance. Indeed, emerging evidence suggests that blood mDNAge inversely associates with grip strength and frailty in ageing humans (Peterson et al., 2022; Verschoor et al., 2021) and is lower following a dietary/physical activity intervention in postmenopausal women (Fiorito et al., 2021). Exercise training and OKSM share substantial gene expression overlap. Given the known phenotypic and functional benefits of exercise adaptation in aged animals (Dungan, Brightwell et al., 2022), an altered transcriptome after brief OKSM-mediated epigenetic partial reprogramming in skeletal muscle could have functional consequences. Prior work shows that OKSM induction in muscle enhances regeneration after injury (Ocampo, Reddy, Martinez-Redondo et al., 2016; Wang et al., 2021), although more work is needed in the context of muscle mass, strength, quality, fatigue resistance and senescent cell abundance with ageing. Exploration into the interaction of epigenetic reprogramming via OKSM in combination with exercise training is also warranted.
Both OKSM and late-life exercise adaption in mice as well as chronic exercise training in humans results in lower levels of Abra/Stars in muscle. Exploratory proteomics recently found that low ABRA/STARS in human skeletal muscle is strongly associated with high physical activity throughout the lifespan (Ubaida-Mohien et al., 2019). ABRA/STARS has numerous functions in muscle (Lamon et al., 2014), but knockdown in myotubes enhances insulin signalling and sensitivity (Jin et al., 2011). Because insulin resistance associates with ageing (Barzilai & Ferrucci, 2012; Fink et al., 1983), lower ABRA/STARS in muscle could be beneficial for improving muscle health throughout the lifespan. Itgb1bp2 (Melusin), Ndufb11 and Romo1 were downregulated by OKSM expression, late-life exercise training and a pulse of MYC in the muscle of mice. Melusin is specific to striated muscle and increases during regeneration (Brancaccio et al., 1999). Conversely, Melusin decreases during atrophy (Vitadello et al., 2020), but its role in exercise adaptation is unclear. ROMO1 generates reactive oxygen species (Chung et al., 2006) and induces senescence (Chung et al., 2008). Downregulation of Romo1 may be beneficial in ageing muscle where senescent cells can manifest and impair adaptation (Dungan, Figueiredo et al., 2022; Dungan, Murach et al., 2022; Zhang, Englund et al., 2022; Zhang, Hong et al., 2022). ROMO1 is also a negative feedback regulator of MYC (Lee et al., 2011), and so its downregulation by MYC induction seems intuitive. Repressed gene expression of the complex I accessory subunit Ndufb11 with OKSM, Myc and exercise training in mice is conserved with chronic exercise in humans. Reduced complex I activity prevents reactive oxygen species production, which enhances cellular fitness and longevity (Rodríguez-Nuevo et al., 2022). Low abundance of specific complex I components including NDUFB11 is a characteristic of tissue from long-lived animals (Miwa et al., 2014; Sahm et al., 2018). Lower Nudfb11 with chronic exercise, probably mediated by MYC, could be part of a larger adaptive response that defends trained skeletal muscle against oxidative damage (Criswell et al., 1993; Parise, Brose et al., 2005; Parise, Phillips et al., 2005; Powers et al., 1999; Radak et al., 1999; Smuder et al., 2011). On balance, studies with ageing and exercise in muscle of humans report a disconnect between mitochondrial gene and protein abundance (Robinson et al., 2017; Tumasian III et al., 2021). Our results should be interpreted with this in mind. Future Ndufb11 gain- and loss-of-function experiments in muscle may be warranted to elucidate its potential role in combatting an aged muscle phenotype.
Myc gene and protein expression is induced in the muscle of young adult humans (Broholm et al., 2011; Brook et al., 2016; Figueiredo et al., 2016; Figueiredo et al., 2021; Popov et al., 2019; Stec et al., 2016; Townsend et al., 2016; Trenerry et al., 2007) and rodents (Armstrong & Esser, 2005; Chen et al., 2002; Goodman et al., 2015; Kirby et al., 2016; Lai et al., 2010; Murach et al., 2022; von Walden et al., 2012; West et al., 2016; Whitelaw & Hesketh, 1992) in response to loading. MYC not only participates in epigenetic reprogramming in concert with Oct3/4, Klf4 and Sox2 (Takahashi & Yamanaka, 2006), but can also facilitate epigenome remodelling on its own (Brenner et al., 2005; Gartel, 2006; Lin et al., 2009; Nakagawa et al., 2010; Pang et al., 2018; Poole et al., 2017; Poole et al., 2019). Consistent with work in other cell types, there was global remodelling of the muscle DNA methylome following MYC induction. We speculate this could be a result of interactions of MYC with DNA methyltransferases and ten eleven translocases (Brenner et al., 2005; Pang et al., 2018; Poole et al., 2019). It is striking that <24 h of MYC induction can change muscle DNA methylation status, but a single bout of exercise also remodels DNA methylation in human muscle tissue (Barres et al., 2012; Maasar et al., 2021; Seaborne et al., 2018). The mechanisms of methylation regulation in muscle by exercise are unclear (Small et al., 2021; Villivalam et al., 2021), but MYC could be a central factor. Recent evidence suggests that overexpression of MYC in aged oligodendrocyte progenitors is sufficient to restore regenerative remyelination in vivo (Neumann et al., 2021); this is consistent with the role of MYC in regulating in vivo sensory nerve regeneration (Ma et al., 2019). Short-term MYC expression recapitulates numerous aspects of myonuclear gene expression at the onset of rapid overload-mediated muscle hypertrophy (Murach et al., 2022). Two weeks of MYC overexpression also mimics the protein synthesis response to exercise-like high-intensity muscle contractions (Mori et al., 2021). Because MYC-controlled gene expression in muscle fibres overlaps with OKSM partial reprogramming and the exercise training-mediated transcriptomes of aged mice, we propose that pulses of Myc could serve to enhance muscle function.
A 60% induction of Myc in the soleus after late-life PoWeR is intriguing because whole-organism MYC knockdown may enhance longevity and healthspan in mice (Hofmann et al., 2015). By contrast, MYC induction in specific cell populations can attenuate cellular ageing and restore regenerative potential (Neumann et al., 2021). Myc gene and protein in muscle tissue may increase in rodents (Hofmann et al., 2014; Mobley et al., 2017) but not humans during ageing (Drummond et al., 2011; Stec et al., 2015). The behaviour of MYC in muscle with ageing is probably complex and could be differentially affected in mononuclear proliferative cells vs. multinuclear post-mitotic muscle fibres within muscle tissue. Single cell RNA-sequencing in muscle revealed Myc enrichment specifically in fibro/adipogenic progenitors, satellite cells, and tenocytes but not myonuclei of aged mice (Zhang, Hong et al., 2022). Elevated Myc in satellite cells of aged mice has been previously described (Price et al., 2014). Cell type-specific upregulation of Myc may explain differing results at the tissue level. Aspects of the gene expression network of Myc, which is operative after muscle contraction when young (Murach et al., 2022; Popov et al., 2019), may be alternatively regulated after resistance training in human muscle when aged (Phillips et al., 2013). MYC gene expression in vastus lateralis muscle samples 4 h after a bout of resistance exercise in ∼80-year-old men and women is blunted (logFC = 0.64 and −0.54, respectively, adjusted P > 0.90) relative to ∼24-year-old men and women (logFC = 1.84 and 1.73, respectively, adjusted P < 0.05, data extracted from MetaMEx) (Pillon et al., 2020; Raue et al., 2012). Blunted MYC gene and protein responsiveness to acute resistance exercise in muscle of younger, albeit still aged humans (∼70 years) has also been observed (Brook et al., 2016; Rivas et al., 2014). Perhaps an attenuated MYC response in muscle fibres of very old humans and animals (Alway, 1997) explains diminished adaptability to exercise, specifically in glycolytic type 2 muscle fibres (Grosicki et al., 2022; Raue et al., 2009; Slivka et al., 2008). We speculate that higher Myc levels in the oxidative soleus muscle after high-volume PoWeR in aged mice is favourable and could contribute to the notable cellular adaptations observed previously (Dungan, Brightwell et al., 2022).
Tighter control over the duration of OKSM and Myc dosing, the timing of sampling after exercise and genetically-induced epigenetic partial reprogramming, the mode of exercise and training status, the potential effects of biological sex, and several other factors may identify a larger gene expression signature of exercise-associated reprogramming-linked genes in muscle. Importantly, understanding whether epigenetic reprogramming, specifically by MYC, induces functional consequences in skeletal muscle is essential. Such insight will help drive healthspan-extending therapeutic efforts forward and advance the ageing field beyond biomarker discovery. Because DNA demethylation is required for cellular reprogramming (Simonsson & Gurdon, 2004), the precise mechanisms by which MYC reshapes the methylome and global epigenome in muscle also deserves further exploration. Ultimately, the mechanisms of muscle molecular reprogramming required to fully recapitulate a youthful phenotype remain elusive, but our data provide a roadmap for further examination of how exercise training combats aspects of ageing. Our data also implicate MYC as an exercise-induced reprogramming factor in skeletal muscle.
Biography
Ronald G. Jones III is a first-year doctoral student working with Dr Kevin A. Murach in the Molecular Muscle Mass Regulation (M3R) Laboratory at the University of Arkansas.
Ronald received his undergraduate degree in Exercise Science from Towson University in Baltimore, Maryland.
He completed a master’s degree in Clinical Exercise Physiology at the University of Delaware.
Ronald is a Certified Clinical Exercise Physiologist under the American College of Sports Medicine and worked in the Baltimore VA Medical Centre as a Research Physiologist. His research focus includes the molecular and cellular mechanisms of skeletal muscle physiology and has a particular interest in -omics.
Originally published at https://physoc.onlinelibrary.wiley.com












