This consulting report was initiated, reviewed (for technical accuracy) and funded by AstraZeneca. BCG was commissioned by AstraZeneca to develop this report.
Key messages
Edited by Joaquim Cardoso MSc*
- To achieve the common goal of all cancer community stakeholders — improved outcomes for people with cancer — sustained collaboration is needed
- … to define the value of oncology medicines in terms of clinical and other value components, including economic benefit and value from the perspective of people with cancer.
- To that end, an international group of leading cancer community experts came together to develop a set of principles for defining and assessing value of cancer therapies
The following seven consensus principles emerged from this expert group:
Principles on oncology-relevant endpoints for consideration in value assessments, particularly for early-stage cancers
- Principle 1: Consider oncology-relevant endpoints other than OS which have intrinsic value for decision-making.
- Principle 2: Continue to build evidence for endpoints that provide earlier indication of treatment efficacy.
- Principle 3: Develop evidence for the next generation of predictive measures that detect and monitor disease.
- Principle 4: Use Managed Entry Agreements (MEA)s supported by ongoing evidence collection to help address decision-maker evidence needs.
Principles on Value Components in Oncology (across all stages of cancer)
- Principle 5: Routinely use PROs in value assessments.
- Principle 6: Assess broad economic impact of new medicines.
- Principle 7: Consider other value aspects of relevance to patients and society.
ORIGINAL PUBLICATION (excerpt)

The Evolving Value Assessment Of Cancer Therapies — Seven Principles from the Cancer Community
Authors
Keith Abrams, Anne-Marie Baird, Susan Brown, Johannes Bruns, Jon Cleland (BCG), Russell Clark, Javier Cortes, Giuseppe Curigliano, Andrea Ferris, Louis P. Garrison, Jens Grueger (BCG), Ataru Igarashi, Hugo Larose (BCG), Myrto Lee (BCG), Gary Lyman, Luca Pani, Zack Pemberton-Whiteley, Tomas Salmonson, Peter Sawicki, Barry Stein, DongChurl Suh, Srikant Vaidyanathan (BCG), Galina Velikova, Richard Vines and Jon Williams (BCG).
November, 2022
The evolving value assessment of cancer therapies: Seven Principles from the Cancer Community
Executive Summary
Cancer management today is moving toward earlier detection and more personalized treatment.
The evolution of oncology science is delivering scientific advancements that can lead to more targeted and effective treatments for people with cancer.
Together, early-stage diagnosis and therapeutic innovation can significantly improve clinical outcomes[1] [2].
Together, early-stage diagnosis and therapeutic innovation can significantly improve clinical outcomes
Investigating and treating cancer earlier has been shown to improve outcomes[3] [4] [5] and early cancer detection has improved in recent decades, owing to a combination of advanced diagnostic technologies and public health campaigns that emphasize the importance of screening and early diagnosis and treatment[6].
As the cancer community looks to leverage scientific innovation to improve outcomes for people with cancer, the identification and utilization of additional oncology-relevant measures should be considered in terms of their role in accelerating the detection of even nascent cancers, speeding up drug development, better informing treatment pathways and value assessments.
As the cancer community looks to leverage scientific innovation to improve outcomes for people with cancer, the identification and utilization of additional oncology-relevant measures should be considered …
… in terms of their role in accelerating the detection of even nascent cancers, speeding up drug development, better informing treatment pathways and value assessments.
People with cancer need access to effective treatment options for them and their particular tumor type in the early stages of their disease to have the optimal chance of transformative and sometimes curative outcomes.
This, in turn, may provide direct and indirect long-term healthcare savings by reducing the amount of healthcare spending per patient[7], limiting the length of time that they are less able to contribute to the workforce and their communities[8] [9], and easing the emotional stress and financial strain of employment-related issues for their caregivers.
The COVID-19 pandemic experience showed how global health systems prioritized investment in healthcare for the benefit of patients, society and the economy.
However, current medicine value assessments do not always consider the full economic and societal benefits of cancer therapies.
The COVID-19 pandemic experience showed how global health systems prioritized investment in healthcare for the benefit of patients, society and the economy.
However, current medicine value assessments do not always consider the full economic and societal benefits of cancer therapies.
To achieve the common goal of all cancer community stakeholders — improved outcomes for people with cancer — sustained collaboration is needed …
… to define the value of oncology medicines in terms of clinical and other value components, including economic benefit and value from the perspective of people with cancer.
To that end, this international group of leading cancer community experts came together to develop a set of principles for defining and assessing value of cancer therapies
(See Section II: Methodology).
We considered: shifts in cancer care toward treating earlier stage disease; enhanced criteria on value principles for the assessment of new medicines, such as those articulated in the value flower from the International Society for Health Economics [Pharmacoeconomics] and Outcomes Research (ISPOR)[10] and constrained resources across the healthcare ecosystem.
Discussions focused on two areas where we agreed that consensus could help shift how the value of medicines is assessed within healthcare systems and processes:
- Oncology-Relevant Endpoints: Which oncology-relevant endpoints should be used for the development and evaluation of treatments for early-stage disease, focusing on breast and lung cancer (two of the mostly commonly studied cancers). Endpoints may be either one or both of 1) predictors (or surrogates) of clinical benefit where correlation is needed to clinical outcomes and 2) with inherent value to people with cancer as an oncology-relevant measure because they will benefit even if it does not show correlation to established endpoints (See Table 1)
- Value Components in Oncology: Which value components are relevant to consider in appraising a medicine. (See Table 2).
Table 1: Oncology-Relevant Endpoints (listed alphabetically)*
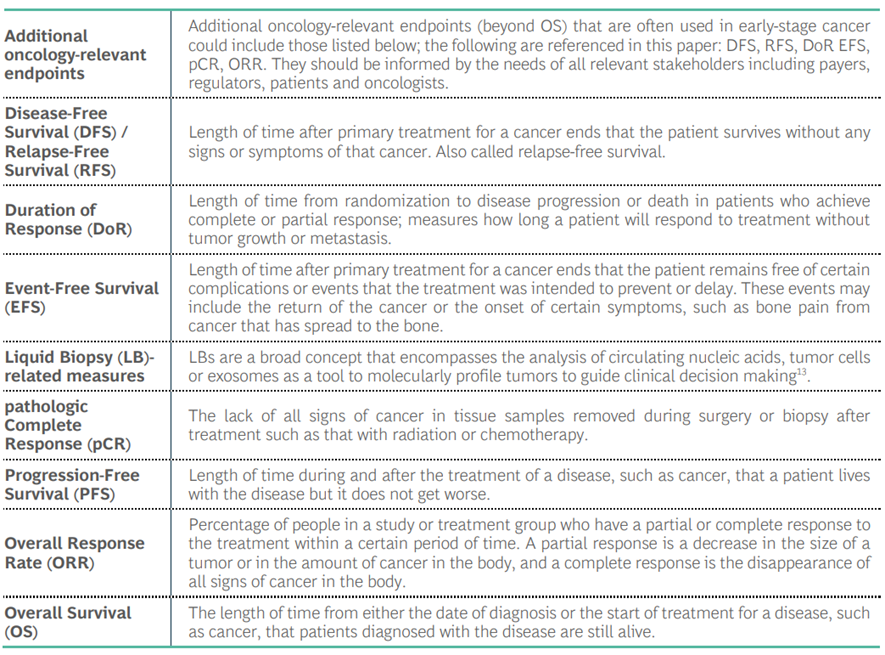
*Unless otherwise noted, definitions are from the National Cancer Institute (NCI) Dictionary of Cancer Terms[11].
Table 2: Value Components in Oncology* (listed alphabetically)
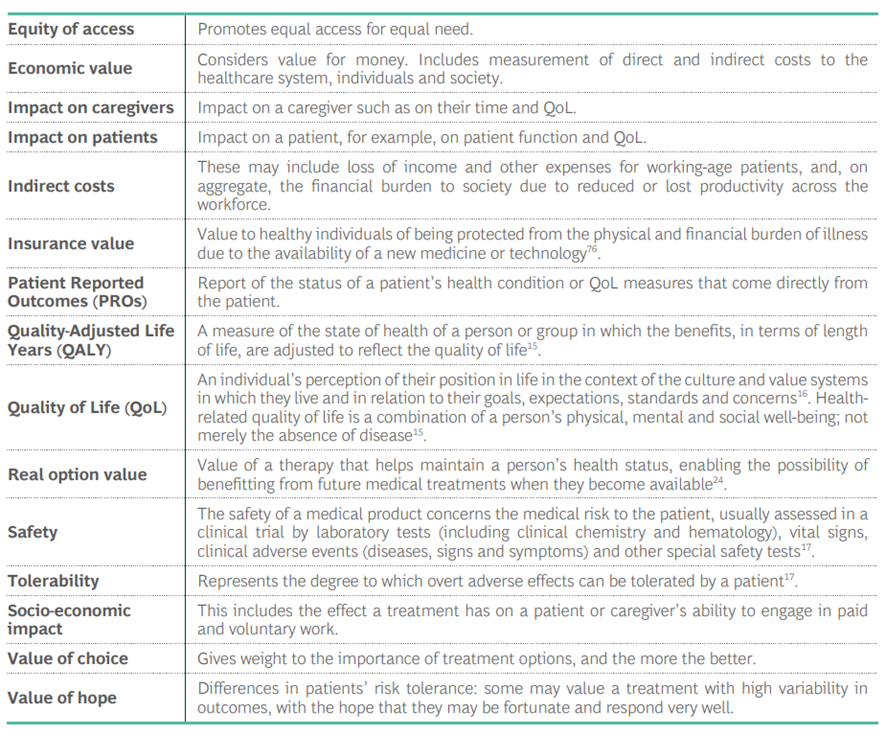
- General definitions of terms shown as used for discussion by this expert group; definitions can vary within the literature.

The following seven consensus principles emerged from this expert group:
Principles on oncology-relevant endpoints for consideration in value assessments, particularly for early-stage cancers
- Principle 1: Consider oncology-relevant endpoints other than OS which have intrinsic value for decision-making.
- Principle 2: Continue to build evidence for endpoints that provide earlier indication of treatment efficacy.
- Principle 3: Develop evidence for the next generation of predictive measures that detect and monitor disease.
- Principle 4: Use Managed Entry Agreements (MEA)s supported by ongoing evidence collection to help address decision-maker evidence needs.
Principles on Value Components in Oncology (across all stages of cancer)
Principle 5: Routinely use PROs in value assessments.
Principle 6: Assess broad economic impact of new medicines.
Principle 7: Consider other value aspects of relevance to patients and society.
Principles on oncology-relevant endpoints for consideration in value assessments, particularly for early-stage cancers
Principle 1: Consider oncology-relevant endpoints other than OS which have intrinsic value for decision-making.
In early-stage cancer OS data takes time to mature or may not be possible to collect in the longer term in early-stage disease.
Indication, intent of treatment and feasibility of measuring patient-relevant outcomes (e.g., EFS, DFS, RFS) within a reasonable timeframe should be evaluated when considering oncology-relevant endpoints as alternative to OS in value assessments.
Principle 2: Continue to build evidence for endpoints that provide earlier indication of treatment efficacy.
Emerging additional oncology-relevant endpoints that can detect treatment impact earlier, such as pCR, generally currently require confirmatory longitudinal outcome data.
As evidence builds that pCR in specific disease settings and therapeutic classes correlates with other outcomes data (e.g., EFS, DFS and RFS), and/or people with cancer and oncologists confirm it reflects meaningful benefit, pCR may become established as a predictor and/or measure of clinical benefit.
Principle 3: Develop evidence for the next generation of predictive measures that detect and monitor disease.
Advances in disease monitoring, such as through circulating tumor DNA (ctDNA), may provide important early information about treatment response and tumor recurrence.
Trials should collect ctDNA data to assess their value as predictors of clinical outcomes.
Principle 4: Use Managed Entry Agreements (MEA)s supported by ongoing evidence collection to help address decision-maker evidence needs.
Carefully designed MEAs with planned confirmatory evidence collection can support timely patient access to new therapeutics and help to address evidence uncertainties associated with earlier access for decision makers.

Principles on Value Components in Oncology (across all stages of cancer)
Principle 5: Routinely use PROs in value assessments.
Data collected from patients via PROs including QoL should be routinely and consistently incorporated into value assessments, along with the value components that are already used relating to safety and efficacy.
Principle 6: Assess broad economic impact of new medicines.
The economic impact of medicines is an essential component of the value assessment and should consider the downstream effect a medicine can have on the amount and associated cost of healthcare resources a patient eventually needs, as well as the socio-economic impact (paid and voluntary work) for patients and those in a caregiving capacity.
Principle 7: Consider other value aspects of relevance to patients and society.
Insurance value, the value of choice, scientific spillovers, equity of access and real option value (See Table 2) should be considered in value assessments, although they may not all be readily quantifiable and may instead require a more qualitative assessment.
This group’s collective interest is the well-being of the millions of people who are diagnosed with cancer every year worldwide.
In 2020 an estimated 19.3 million people were diagnosed with cancer globally which included an estimated 4.5 million for breast and lung cancers alone[12].
To benefit from advancements in early detection and treatment, people with cancer need timely access to medicines for early-stage disease when treatment can be most impactful[13].
To benefit from advancements in early detection and treatment, people with cancer need timely access to medicines for early-stage disease when treatment can be most impactful.
A list of recommendations is included in Section V: Next Steps.
Bringing together different voices is a powerful way to stimulate discussion and kick-start a cross-healthcare ecosystem effort that could generate the changes needed to benefit people with cancer, those in a caregiving capacity and society as a whole.
Voices of people with cancer are essential to the development of value assessments in order to better, and more holistically, understand the impact of cancer and its treatment on people with cancer.
This group’s collective interest is the well-being of the millions of people who are diagnosed with cancer every year worldwide.
In 2020 an estimated 19.3 million people were diagnosed with cancer globally which included an estimated 4.5 million for breast and lung cancers alone[14].

Methodology [excerpt from the long version]
A Delphi-based process to build consensus across the cancer community
This expert group represents a range of professionals in the cancer community:
patient advocates, oncologists with different specializations, health economists, regulators, members of payer and HTA bodies and representatives from professional societies. The insights and ideas contributed are individual, and not of the organizations represented, past or present. Additionally, only those who had previously worked in regulatory agencies, payers and HTA bodies were involved to protect against any conflict of interest.
Discussions followed the Delphi approach to build consensus across a range of expertise and topics[1].
Over five months, sixteen members of the expert group participated in numerous structured interactions, including a survey, two virtual plenary meetings (panels), one-on-one interviews and structured discussion on a secure social platform.
Eight additional experts contributed their perspectives through individual interviews.
The group (hereafter “we”) represented perspectives from Australia, Canada, China, France, Germany, Italy, Japan, South Korea, Singapore, Spain, Sweden, the UK and US.
We discussed two areas where consensus could help shift how the value of treatments is assessed:
(1) which oncology-relevant endpoints to use for assessing the benefit of treatments for early-stage cancer in clinical trials, and access decisions for early-stage cancer treatments and
(2) which additional value components are important and how they can be integrated in value assessments within the healthcare system and processes.
We did not discuss pricing of medicines.
Our research and discussions were guided by the universal recognition that cancer care is moving to
- focus on treatment of early-stage disease and that
- accelerating development and regulatory approval for medicines in this setting has implications for the maturity of the evidence available for access and funding decision-making.
References and Additional Information
See the original publication
https://media-publications.bcg.com
Authors
Keith Abrams, Anne-Marie Baird, Susan Brown, Johannes Bruns, Jon Cleland (BCG), Russell Clark, Javier Cortes, Giuseppe Curigliano, Andrea Ferris, Louis P. Garrison, Jens Grueger (BCG), Ataru Igarashi, Hugo Larose (BCG), Myrto Lee (BCG), Gary Lyman, Luca Pani, Zack Pemberton-Whiteley, Tomas Salmonson, Peter Sawicki, Barry Stein, DongChurl Suh, Srikant Vaidyanathan (BCG), Galina Velikova, Richard Vines and Jon Williams (BCG).
Acknowledgments
Project Support
The authors would like to acknowledge and thank the following BCG project support, whose expertise and guidance supported the planning phase, data collection, report write-up and final editorial review stage (in alphabetical order): Anni Ding, Aparna Shekar, Annabelle Sherwood and Ashley Veselik. Cheryl de Jong-Lambert, medical writer, also contributed to report.
Cancer Community Experts
The international expert group (hereafter “expert group”) brought expertise from around the healthcare ecosystem in cancer care. The experts contributed their time and expertise to the key themes described herein and the authors (above) have reviewed the report. They included patient advocates, payers, health economists, regulators, professional oncology societies and physicians, and represented perspectives from Australia, Canada, China, France, Germany, Italy, Japan, South Korea, Singapore, Spain, Sweden, the UK and US. The insights, ideas and key themes contributed are individual, and not of the organizations represented, past or present.
Sixteen experts participated as panelists across numerous fora over five months and are among the authors of this paper (See above): Keith Abrams, Susan Brown, Johannes Bruns, John Carpten, Russell Clark, Javier Cortes, Giuseppe Curigliano, Andrea Ferris, Louis P. Garrison, Gary Lyman, Luca Pani, Zack Pemberton-Whiteley, Bettina Ryll, Tomas Salmonson, Peter Sawicki and Richard Vines. An additional eight representatives were interviewed outside the panel group: Anne-Marie Baird, Y. K. Gupta, Ataru Igarashi, Ravindran Kanesvaran, Zhao Kun, Barry Stein, Dong-Churl Suh and Galina Velikova.
Commissioning, Review and Funding by AstraZeneca
This consulting report was initiated, reviewed (for technical accuracy) and funded by AstraZeneca. BCG was commissioned by AstraZeneca to develop this report. Some experts were provided compensation for their time in participating, and some experts chose to forego compensation.












